We are working on a drug discovery based on structural analysis using a Cryo-electron microscope (Cryo-EM).

What is a Cryo-EM?
The Cryo-electron microscope (Cryo-EM) Single Particle Analysis (SPA) method is one of the structural analysis methods using an electron microscope, which was awarded the 2017 Chemistry Nobel Prize and has been receiving a lot of attention in recent years.
Conventional drug discovery research (HTS) requires millions of trials and errors (evaluation of millions of compounds) until a hit appears against a protein of unknown structure. This approach requires cost and time.
By using a Cryo-EM, structural analysis became possible for many proteins that were difficult to analyze by X-ray structure analysis, which is known as a conventional analysis method. By introduction of Cryo-EM, the number of proteins that can be reliably analyzed has increased, and it is paving the way in expanding a new drug discovery area.

Currently, we are conducting drug discovery research programs using several Cryo-EMs, Titan Krios G4,Talos Arctica G2 and Titan Krios G3i manufactured by Thermo Fisher Scientific .
-

Talos Arctica G2 -

Titan Krios G3i -

Titan Krios G4
Types of electron microscopes
 Transmission Electron Microscopy (TEM)
Transmission Electron Microscopy (TEM)
- Apply an electron beam and observe the transmitted electrons
- The density of transmitted electrons changes depending on the components of the sample
- The electron beam wavelength of the acceleration voltage of 300 kV is 0.002 nm.
 Scanning Electron Microscopy(SEM)
Scanning Electron Microscopy(SEM)
- Scan the sample surface with an electron beam
- Create an image based on the signals of secondary electrons and backscattered electrons
![]()

Cryogenic Electron Microscopy
Cryogenic is a technology that generates low temperature. The term comes from combination of “cryos” which means “cold” and “gennese” which means “to produce” in Greek. Cryogenic electron microscope, commonly known as CryoEM, is a transmission electron microscope (transmission electron microscope) that irradiates biomolecules such as proteins with electron beams under liquid nitrogen (-196 ° C) cooling and observes samples. This is a new method based on TEM). As a method for determining the three-dimensional structure of proteins with high resolution, we have made remarkable technological innovations in detectors, etc. In 2017, the Nobel Prize in Chemistry was awarded to three overseas researchers who contributed to the development. In 1995, it was theoretically shown that this new method can analyze the structure of molecules up to 100 kDa * ¹, and since then, especially after 2015, active research and development has been carried out, and the recent “Resolution Revolution” makes it possible to elucidate the molecular structure with a resolution of 1.5 Å * ² or less. Recently, it has become possible to analyze the structure of molecules in the vicinity of about 50 to 60 kDa. ![]()
By resolution: Changes in the number of protein analyzes by CryoEM
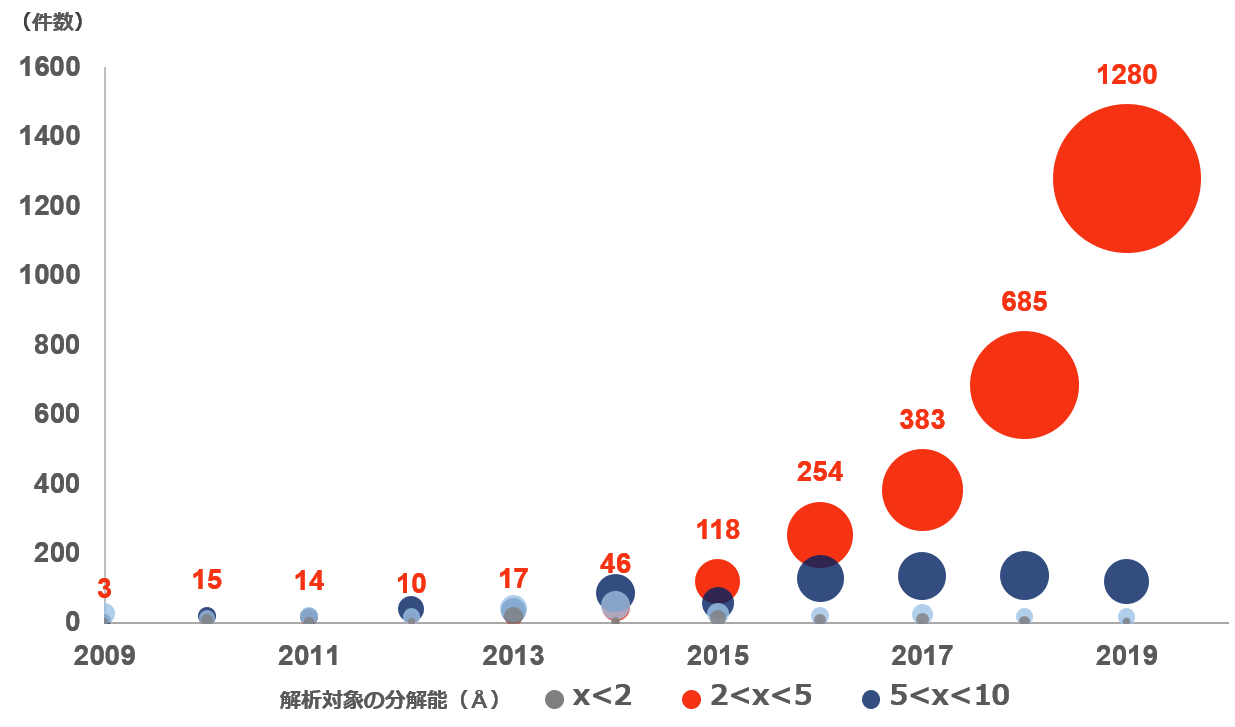 It can be confirmed that the number of cases with a resolution of 2Å-5Å has increased sharply in the past 5 years. Source: Created by information from Protein Data Bank Japan
It can be confirmed that the number of cases with a resolution of 2Å-5Å has increased sharply in the past 5 years. Source: Created by information from Protein Data Bank Japan
- *¹ Da (Dalton): 1/12 of the mass of the 12C atom, which is a unit expressing mass, is 1. kDa (kilodalton)
- *² Å (Angstrom): 1 Å is defined as 10-10m = 0.1 nanometer (nm) = 100 picometers (pm)
Single particle analysis method What is Single Particle Analysis (SPA)? A large number of images (photographs) of protein molecules are taken using a transmission electron microscope under liquid nitrogen temperature conditions (cryo). It is possible to reduce noise by taking the averaging of a large number of captured images, and improve the resolution of the observed protein through averaging in 2D and averaging in 3D construction.
Strengths of Cryo-EM Single Particle Analysis method
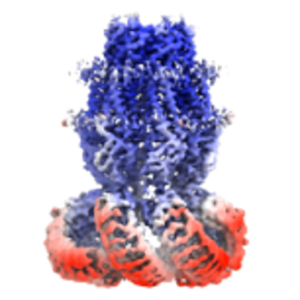
Features and benefits of CryoEM
- No need to crystallize target
- Maintaining an environment close to the living body
- Supports large molecules containing multiple subunits
- Structural observation at the atomic level is possible
- Experimental conditions can be changed according to the functional status
Strengths and areas of expertise in drug discovery
- Structural analysis of targets is possible in an environment close to the living body
- Complexes and transmembrane proteins that were difficult to crystallize can also be observed
- Allows structural analysis of proteins that are yet unrevealed

